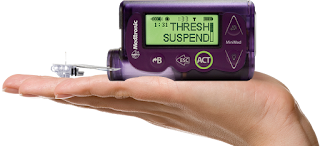
DAILY INNOVATION BRIEF by Maryanne Kane, Journalist

D AILY INNOVATION BRIEF By Journalists Edward Kane & Maryanne Kane BMW's VERY SMART CAR Source: BMW BMW'S new 5 Series Saloon is so smart it suggests when to change lanes and, with the driver's approval, changes lanes on its own Driver agrees with the lane cha...